By Katie Marie
Why Hydrogen?
Hydrogen can replace fossil fuels in essentially any area. It can be processed into fuel for airplanes and ships. It can be burned to generate electricity, power cars or facilitate industrial processes that require a large amount of heat. Factors related to the storage, transport and safety of hydrogen, as well as how it compares economically with other fossil fuel replacements, will determine where it becomes common.

Like natural gas and oil products, hydrogen can be burned to produce heat or electricity. Unlike other fuels that produce CO2 when they combust, hydrogen reacts with oxygen in the air to produce H2O — good old water.
For hydrogen to serve as a climate solution rather than a problem, it must be produced using renewable energy or fossil fuels offset by carbon capture and storage systems. At present, producing green hydrogen from renewable energy sources is expensive, which is why 95% of hydrogen globally is produced from non-renewable fossil sources.
Nageh Allam, professor of physics and head of AUC’s Energy Materials Lab, has been researching ways to improve the efficiency and reduce the cost of green hydrogen production by substituting new materials in the process.
The Catalyst
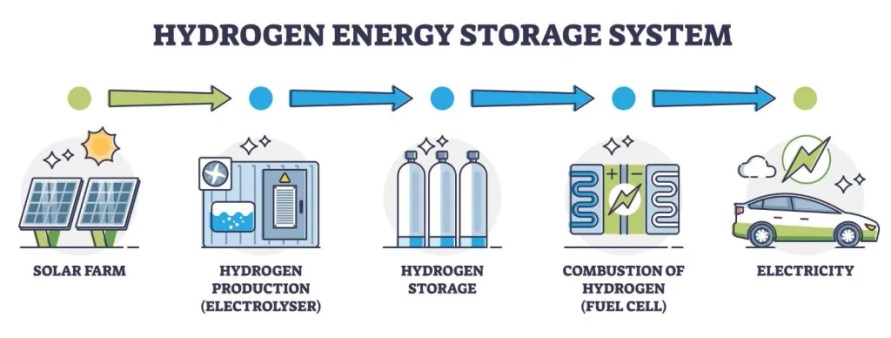
Green hydrogen is produced through a process called electrolysis, where electrical energy, usually supplied from a renewable power plant, is used to split water molecules. “Water on its own will never absorb a sufficient amount of energy to split, so we need to use a material that we call a ‘catalyst’ to make this happen,” Allam explained. “Right now, the main catalyst used in electrolysis is platinum. It’s a highly efficient metal but also very rare. It’s expensive and difficult to source, and this is a big part of what makes green hydrogen production costly.”
Allam has two active projects experimenting with new catalysts in green hydrogen production. One is focused on the traditional method in which electrolysis is powered by electricity from a renewable power plant.
“We are finding lower-cost materials that can be used as catalysts and still achieve a degree of efficiency close to that of existing commercial electrolyzers,” he said. “In particular, we are concentrating on a selection of ‘transition metals’ that are relatively abundant in the Earth’s crust. To further reduce the cost, we’re obtaining them from recycled materials.”
As devices like computers and mobile phones have become a core part of our daily lives, humanity has begun to generate significant quantities of electronic waste. “Tech stores will often give away old electronics for free,” Allam said. “In our lab, we separate out the metallic parts, crush them into a powder then use a process called hydrometallurgy to isolate the ones we need.”
Allam’s other green hydrogen project involves experimentation with different compositions of semiconductors as catalysts in solar-assisted electrolysis. This is a less mature technology for hydrogen production whereby the catalyst absorbs sunlight directly to split water molecules rather than deriving its energy from a power plant. Currently, the efficiency of this process is very low, around 3% on average, and again Allam is seeking to identify a catalyst that is stable, affordable and efficient.
“To give you an example of the tradeoff, iron is cheap and can absorb an enormous amount of sunlight, but it also corrodes easily,” Allam noted. “When it comes into contact with water during electrolysis, it dissolves rapidly over time. One approach we’re interested in is adding different materials to iron to make it more stable.”
Egypt’s Ambitions
As part of its own energy transition, Egypt is seeking to become a regional hub for green energy, and the government sees significant potential in the area of green hydrogen. A committee is set to unveil a $40 billion national hydrogen strategy with a target output of 1.4 gigawatts by 2030.
In the last year, the government has actively solicited proposals for small-scale, green hydrogen production pilot projects and signed memoranda of understanding with companies such as Germany’s Siemens Energy, Éléctricité de France and Denmark’s Maersk.
With all of this new momentum, Allam hopes his research can play a meaningful role in furthering Egypt’s green hydrogen ambitions. “Foreign companies in Egypt will be installing ready-made systems, but when you bring technology to a new setting, there are always challenges,” he said. “For example, dust causes problems with solar panels in Egypt in a way that it does not in Europe and the United States. Or there could be a problem with the composition of water in Egypt that impacts imported electrolyzers. Maybe our research will lead to a better catalyst or one that is more durable in the Egyptian climate. In general, our lab could provide research and development for companies coming into the country. We know the environment as well as the technology.”




Leave a Reply